搜索结果: 'methocult media formulations for mouse hematopoietic cells serum containing'
-
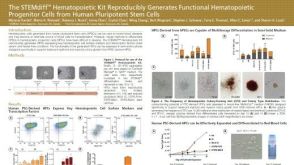 科学海报The STEMdiff™ Hematopoietic Kit Reproducibly Generates Functional Hematopoietic Progenitor Cells from Human Pluripotent Stem Cells
科学海报The STEMdiff™ Hematopoietic Kit Reproducibly Generates Functional Hematopoietic Progenitor Cells from Human Pluripotent Stem Cells产品类型:
Conference:
ISEH 2017
产品号#:
05310
04636
产品名:
STEMdiff™ 造血试剂盒
MethoCult™ SF H4636
-
 科学海报Stroma-Free, Serum-Free Expansion and Differentiation of Hematopoietic Stem and Progenitor Cells to the T Cell Lineage
科学海报Stroma-Free, Serum-Free Expansion and Differentiation of Hematopoietic Stem and Progenitor Cells to the T Cell Lineage产品类型:
Conference:
AAi 2017
产品号#:
09605
09900
09910
09920
产品名:
StemSpan™ SFEM II
-
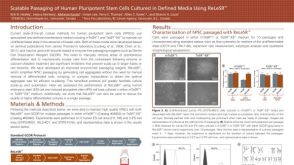 科学海报Scalable Passaging of Human Pluripotent Stem Cells Cultured in Defined Media Using ReLeSR™
科学海报Scalable Passaging of Human Pluripotent Stem Cells Cultured in Defined Media Using ReLeSR™产品类型:
Conference:
PLURIMES 2014
产品号#:
05872
05873
07174
产品名:
-
 技术公告Isolate Mouse CD45.1 or CD45.2 Positive Cells with EasySep™ Release Mouse Positive Selection Kits
技术公告Isolate Mouse CD45.1 or CD45.2 Positive Cells with EasySep™ Release Mouse Positive Selection Kits产品类型:
细胞类型:
B细胞,NK细胞,T细胞,先天性淋巴细胞,单个核细胞,单核细胞,巨噬细胞,树突状细胞(DCs),浆细胞,淋巴细胞,白细胞,粒细胞及其亚群,调节性T细胞,造血干祖细胞,髓系细胞
产品号#:
产品名:
发布日期: 01/15/2025 -
 技术公告Generation of T Cells from Human Pluripotent Stem Cells Using STEMdiff™ and StemSpan™ Media and Supplements
技术公告Generation of T Cells from Human Pluripotent Stem Cells Using STEMdiff™ and StemSpan™ Media and Supplements产品类型:
产品号#:
产品名:
发布日期: 10/17/2023


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒







 沪公网安备31010102008431号
沪公网安备31010102008431号