搜索结果: 'methocult media formulations for human hematopoietic cells serum containing'
-
 科学海报An Animal Component-Free, Serum-Free Culture Method for Generation of Human Dendritic Cells
科学海报An Animal Component-Free, Serum-Free Culture Method for Generation of Human Dendritic Cells产品类型:
Conference:
AAI 2016
产品号#:
10985
10986
10987
10988
10989
产品名:
ImmunoCult™ 树突状细胞培养试剂盒
ImmunoCult™-ACF树突状细胞培养基
ImmunoCult™-ACF树突状细胞培养基
ImmunoCult™-ACF树突状细胞分化添加物
ImmunoCult™树突状细胞成熟添加物
发布日期: 07/20/2016 -
 技术公告Generation of T Cells from Human Pluripotent Stem Cells Using STEMdiff™ and StemSpan™ Media and Supplements
技术公告Generation of T Cells from Human Pluripotent Stem Cells Using STEMdiff™ and StemSpan™ Media and Supplements产品类型:
产品号#:
产品名:
发布日期: 10/17/2023 -
 技术公告Flow Cytometry Methods for Identifying Mouse Hematopoietic Stem and Progenitor Cells
技术公告Flow Cytometry Methods for Identifying Mouse Hematopoietic Stem and Progenitor Cells产品类型:
细胞类型:
造血干祖细胞
产品号#:
产品名:
发布日期: 08/31/2018 -
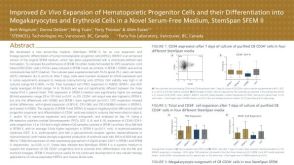 科学海报Improved Ex Vivo Expansion of Hematopoietic Progenitor Cells from Cord Blood in a Novel Serum and Animal Component Free Medium
科学海报Improved Ex Vivo Expansion of Hematopoietic Progenitor Cells from Cord Blood in a Novel Serum and Animal Component Free Medium产品类型:
Conference:
ISCT 2012
产品号#:
产品名:
-

产品类型:
产品号#:
04436
04064
04100
04230
04236
04431
04434
04444
04464
04531
04535
04545
04536
04564
04035
04330
04034
04044
04435
04445
04534
04544
04437
04447
产品名:
MethoCult™ SF H4436
MethoCult™ H4034 Optimum 入门试剂盒
MethoCult™ H4100
MethoCult™ H4230
MethoCult™ SF H4236
MethoCult™ H4431
MethoCult™ H4434 Classic
MethoCult™ H4434 Classic
MethoCult™ H4434 Classic 套装
MethoCult™ H4531
MethoCult™ H4535 Enriched,不含EPO
MethoCult™ H4535 Enriched,不含EPO
MethoCult™ SF H4536
MethoCult™ H4534 Classic 无 EPO 入门试剂盒
MethoCult™ 不含EPO的H4035 Optimum
MethoCult™ H4330
MethoCult™ H4034 Optimum
MethoCult™ H4034 Optimum
MethoCult™ H4435 Enriched
MethoCult™ H4435 Enriched
MethoCult™ H4534 Classic(不含 EPO)
MethoCult™ H4534 Classic(不含 EPO)
MethoCult™ Express
MethoCult™ Express


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒






 沪公网安备31010102008431号
沪公网安备31010102008431号