技术资料
-
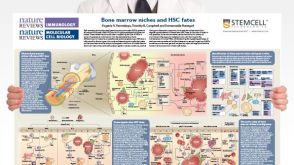 挂图Bone Marrow Niches and HSC Fates A detailed reference on signaling pathways in the bone marrow and how these influence HSC fate decisions; created in partnership with Nature Reviews Immunology and Nature Reviews Molecular Cell Biology
挂图Bone Marrow Niches and HSC Fates A detailed reference on signaling pathways in the bone marrow and how these influence HSC fate decisions; created in partnership with Nature Reviews Immunology and Nature Reviews Molecular Cell Biology -
 46:01
线上讲座Targeting Self-Renewal Function in Normal Hematopoietic and Leukemic Stem Cells发布日期: 02/03/2017
46:01
线上讲座Targeting Self-Renewal Function in Normal Hematopoietic and Leukemic Stem Cells发布日期: 02/03/2017
过滤器
筛选结果
细胞类型
- B 细胞 213 项目
- CD4+ 3 项目
- CD4+ T细胞 132 项目
- CD8+ 3 项目
- CD8+ T细胞 108 项目
- CHO细胞 15 项目
- HEK-293细胞(人胚肾293细胞) 2 项目
- NK 细胞 161 项目
- PSC衍生 32 项目
- T 细胞 385 项目
- 上皮细胞 142 项目
- 中胚层 3 项目
- 乳腺细胞 91 项目
- 先天性淋巴细胞 30 项目
- 全血 15 项目
- 其他细胞系 10 项目
- 内皮细胞 7 项目
- 内胚层 2 项目
- 前列腺细胞 18 项目
- 单个核细胞 88 项目
- 单核细胞 169 项目
- 多发性骨髓瘤细胞 6 项目
- 多能干细胞 1895 项目
- 小胶质细胞 13 项目
- 巨噬细胞 43 项目
- 巨核细胞 10 项目
- 心肌细胞 19 项目
- 成骨细胞 10 项目
- 星形胶质细胞 14 项目
- 杂交瘤细胞 91 项目
- 树突状细胞(DCs) 115 项目
- 淋巴细胞 72 项目
- 癌细胞及细胞系 149 项目
- 白细胞 27 项目
- 白细胞单采样本 16 项目
- 白血病/淋巴瘤细胞 15 项目
- 监管 1 项目
- 真皮细胞 3 项目
- 神经元 1 项目
- 神经干/祖细胞 444 项目
- 神经细胞 9 项目
- 粒细胞及其亚群 89 项目
- 红系细胞 12 项目
- 红细胞 13 项目
- 肌源干/祖细胞 11 项目
- 肝细胞 37 项目
- 肠道细胞 103 项目
- 肾细胞 4 项目
- 肿瘤细胞 28 项目
- 胰腺细胞 17 项目
- 脂肪细胞 6 项目
- 脑肿瘤干细胞 103 项目
- 血小板 4 项目
- 血管生成细胞 1 项目
- 角质形成细胞 1 项目
- 软骨细胞 9 项目
- 造血干/祖细胞 906 项目
- 造血干祖细胞 3 项目
- 造血细胞 1 项目
- 间充质基质细胞 26 项目
- 间充质干/祖细胞 148 项目
- 间充质干祖细胞 1 项目
- 骨髓基质细胞 2 项目
- 骨髓间质细胞 1 项目
- 髓系细胞 134 项目
- 肾脏细胞 8 项目
- CD4+T细胞 1 项目
- CD8+T细胞 1 项目
- PSC衍生上皮细胞 48 项目
- PSC衍生中胚层 29 项目
- PSC衍生内皮细胞 27 项目
- PSC衍生内胚层 32 项目
- PSC衍生心肌细胞 32 项目
- PSC衍生神经细胞 139 项目
- PSC衍生肝细胞 24 项目
- PSC衍生造血干细胞 47 项目
- PSC衍生间充质细胞 32 项目
- 其他T细胞亚型 32 项目
- 呼吸道细胞 101 项目
- 多巴胺能神经元 7 项目
- 小鼠胚胎成纤维细胞 1 项目
- 浆细胞 19 项目
- 神经元 200 项目
- 调节性T细胞 70 项目
Show More
Show Less
研究领域
- HIV 72 项目
- HLA 52 项目
- 上皮细胞生物学 273 项目
- 免疫 20 项目
- 免疫学 892 项目
- 呼吸系统研究 52 项目
- 嵌合体 24 项目
- 干细胞生物学 2728 项目
- 感染性疾病(传染病) 1 项目
- 抗体制备 7 项目
- 新陈代谢 7 项目
- 杂交瘤制备 2 项目
- 疾病建模 259 项目
- 神经科学 635 项目
- 移植研究 102 项目
- 类器官 199 项目
- 细胞外囊泡研究 10 项目
- 细胞治疗开发 1 项目
- 细胞疗法开发 145 项目
- 细胞系制备 194 项目
- 脐带血库 61 项目
- 药物发现和毒理检测 401 项目
- 血管生成细胞研究 1 项目
- 传染病 84 项目
- 内皮细胞生物学 8 项目
- 杂交瘤生成 15 项目
- 癌症研究 679 项目
- 血管生成细胞研究 43 项目
Show More
Show Less
产品系列
- ALDECOUNT 9 项目
- CellPore 1 项目
- ES-Cult 3 项目
- HetaSep 1 项目
- MegaCult 32 项目
- ALDEFLUOR 61 项目
- AggreWell 4 项目
- ArciTect 3 项目
- ClonaCell 1 项目
- CryoStor 5 项目
- EasySep 25 项目
- HemaTox 2 项目
- ImmunoCult 3 项目
- MesenCult 4 项目
- MethoCult 408 项目
- MyeloCult 22 项目
- RSeT 1 项目
- RosetteSep 4 项目
- STEMdiff 11 项目
- STEMvision 4 项目
- StemSpan 161 项目
- TeSR 4 项目
Show More
Show Less
资源类别
- 专家访谈 3 项目
- 产品手册 22 项目
- 实验方案 5 项目
- 技术公告 10 项目
- 技术窍门 8 项目
- 挂图 4 项目
- 播客 1 项目
- 文献 696 项目
- 点播课程 1 项目
- 科学海报 15 项目
- 线上讲座 16 项目
- 视频 13 项目
Show More
Show Less


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒





 沪公网安备31010102008431号
沪公网安备31010102008431号