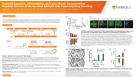
Pancreatic β cells derived from human induced pluripotent stem cells (hiPSCs) represent a promising therapeutic avenue in regenerative medicine for diabetes treatment. However,current differentiation protocols lack the specificity and efficiency required to reliably produce fully functional β cells,limiting their clinical applicability. Epigenetic barriers,such as histone modifications,may hinder proper differentiation and the acquisition of essential maturation markers in these cells. Methods: hiPSCs were cultured under feeder-free conditions and subjected to lentiviral transduction with shRNA constructs to silence KDM4A. Differentiation into pancreatic β-like cells was performed using stepwise protocols,with or without doxycycline supplementation,to evaluate the effect of KDM4A suppression. Gene expression was quantified by RT-qPCR,protein expression was assessed by western blotting and immunofluorescence,and functional insulin release was determined by glucose-stimulated insulin secretion (GSIS) assays. Statistical analysis was conducted using unpaired two-tailed Student’s t-tests,with significance set at p < 0.05. Results: A reduction in pancreatic development proteins was observed in the different differentiation states evaluated,after blocking KDM4A expression. Knockdown of KDM4A significantly reduced the expression of pancreatic β-cell genes,such as PDX1,Nkx6.1,and Ins,by 50% compared to WT iPSCs differentiated under the same conditions. Similarly,glucose-stimulated insulin secretion was reduced by approximately 80% in KDM4A-deficient β-like cells. Conclusions: These results emphasize the critical role of histone demethylation in hiPSC differentiation toward β cells. Our findings identify KDM4A as a key epigenetic regulator,suggesting that its modulation could enhance the generation of functional β cells for regenerative medicine in diabetes.
View Publication
 实验方案Differentiation of hPSCs to Ventricular Cardiomyocytes in 3D Culture
实验方案Differentiation of hPSCs to Ventricular Cardiomyocytes in 3D Culture

 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒










 沪公网安备31010102008431号
沪公网安备31010102008431号