技术资料
-
 实验方案Optimizing Delivery Efficiency with Fluorescent Dextran Using the CellPore™ Transfection System
实验方案Optimizing Delivery Efficiency with Fluorescent Dextran Using the CellPore™ Transfection System研究方向:
免疫学,疾病建模,药物发现和毒性检测
-
 技术公告Uniform and Scalable EB Formation for Drug Discovery and Regenerative Medicine Applications
技术公告Uniform and Scalable EB Formation for Drug Discovery and Regenerative Medicine Applications细胞类型:
PSC衍生造血细胞,多能干细胞,PSC衍生内胚层,PSC衍生心肌细胞 ,PSC衍生神经细胞
发布日期: 06/01/2012 -
 实验方案Gene Editing Human Pluripotent Stem Cells (hPSCs) Using the CellPore™ Transfection System
实验方案Gene Editing Human Pluripotent Stem Cells (hPSCs) Using the CellPore™ Transfection System研究方向:
免疫学,疾病建模,药物发现和毒性检测
-
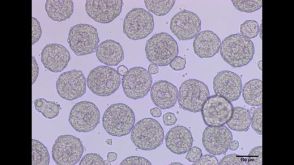 实验方案Differentiation of hPSCs to Ventricular Cardiomyocytes in 3D Culture
实验方案Differentiation of hPSCs to Ventricular Cardiomyocytes in 3D Culture研究方向:
干细胞生物学,疾病建模,细胞治疗开发,细胞系制备,药物发现和毒性检测


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒










 沪公网安备31010102008431号
沪公网安备31010102008431号