技术资料
-
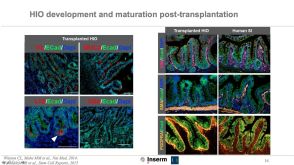
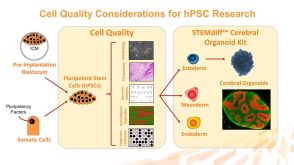
 科学海报A Reliable, Efficient, and Matrix-Free Method to Generate Midbrain Organoids from Human Pluripotent Stem Cells
科学海报A Reliable, Efficient, and Matrix-Free Method to Generate Midbrain Organoids from Human Pluripotent Stem CellsConference:
CSHL 3D Brain 2022
-
 43:00
线上讲座Cerebral Organoids as 3D, Stem Cell-Derived Models of Tuberous Sclerosis Complex发布日期: 04/07/2020
43:00
线上讲座Cerebral Organoids as 3D, Stem Cell-Derived Models of Tuberous Sclerosis Complex发布日期: 04/07/2020 -
 47:18
线上讲座Madeline Lancaster on Brain Organoids: Modeling Human Brain Development in a Dish发布日期: 09/20/2017
47:18
线上讲座Madeline Lancaster on Brain Organoids: Modeling Human Brain Development in a Dish发布日期: 09/20/2017


 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒










 沪公网安备31010102008431号
沪公网安备31010102008431号