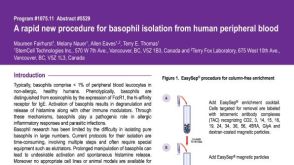
Basophils (Ba) and mast cells (MC) are important effector cells of inflammatory reactions. Both cell types derive from CD34(+) hematopoietic progenitors. However,little is known about the cell subsets that become committed to and give rise to Ba and/or MC. We have generated a monoclonal antibody (MoAb),97A6,that specifically detects human Ba,MC (lung,skin),and their CD34(+) progenitors. Other mature hematopoietic cells (neutrophils,eosinophils,monocytes,lymphocytes,platelets) did not react with MoAb 97A6,and sorting of 97A6(+) peripheral blood (PB) and bone marrow (BM) cells resulted in an almost pure population (textgreater98%) of Ba. Approximately 1% of CD34(+) BM and PB cells was found to be 97A6(+). Culture of sorted CD34(+)97A6(+) BM cells in semisolid medium containing phytohemagglutinin-stimulated leukocyte supernatant for 16 days (multilineage assay) resulted in the formation of pure Ba colonies (10 of 40),Ba-eosinophil colonies (7 of 40),Ba-macrophage colonies (3 of 40),and multilineage Ba-eosinophil-macrophage and/or neutrophil colonies (12 of 40). In contrast,no Ba could be cultured from CD34(+)97A6(-) cells. Liquid culture of CD34(+) PB cells in the presence of 100 ng/mL interleukin (IL)-3 (Ba progenitor assay) resulted in an increase of 97A6(+) cells,starting from 1% of day-0 cells to almost 70% (basophils) after day 7. Culture of sorted BM CD34(+)97A6(+) cells in the presence of 100 ng/mL stem cell factor (SCF) for 35 days (mast cell progenitor assay) resulted in the growth of MC (textgreater30% on day 35). Anti-IgE-induced IgE receptor cross-linking on Ba for 15 minutes resulted in a 4-fold to 5-fold upregulation of 97A6 antigen expression. These data show that the 97A6-reactive antigen plays a role in basophil activation and is expressed on multipotent CD34(+) progenitors,MC progenitors,Ba progenitors,as well as on mature Ba and tissue MC. The lineage-specificity of MoAb 97A6 suggests that this novel marker may be a useful tool to isolate and analyze Ba/MC and their progenitors.
View Publication
 科学海报Isolation of Tumor-Infiltrating Leukocytes from Mouse Tumors
科学海报Isolation of Tumor-Infiltrating Leukocytes from Mouse Tumors 技术公告Achieve Scalable, High-Quality Nucleic Acid Extractions with the EasySep™ Total Nucleic Acid Extraction Kit
技术公告Achieve Scalable, High-Quality Nucleic Acid Extractions with the EasySep™ Total Nucleic Acid Extraction Kit

 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒








 沪公网安备31010102008431号
沪公网安备31010102008431号