技术资料
-
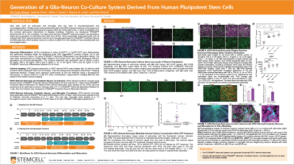 科学海报Generation of a Glia-Neuron Co-Culture System Derived From Human Pluripotent Stem Cells
科学海报Generation of a Glia-Neuron Co-Culture System Derived From Human Pluripotent Stem CellsConference:
GLIA 2021
-
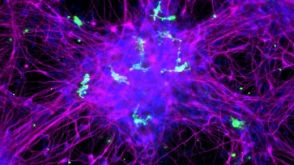 实验方案How to Co-Culture Human Pluripotent Stem Cell (hPSC)-Derived Forebrain Neurons and Microglia
实验方案How to Co-Culture Human Pluripotent Stem Cell (hPSC)-Derived Forebrain Neurons and Microglia研究方向:
免疫,疾病建模,神经科学,传染病


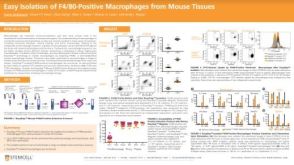
 EasySep™小鼠TIL(CD45)正选试剂盒
EasySep™小鼠TIL(CD45)正选试剂盒













 沪公网安备31010102008431号
沪公网安备31010102008431号